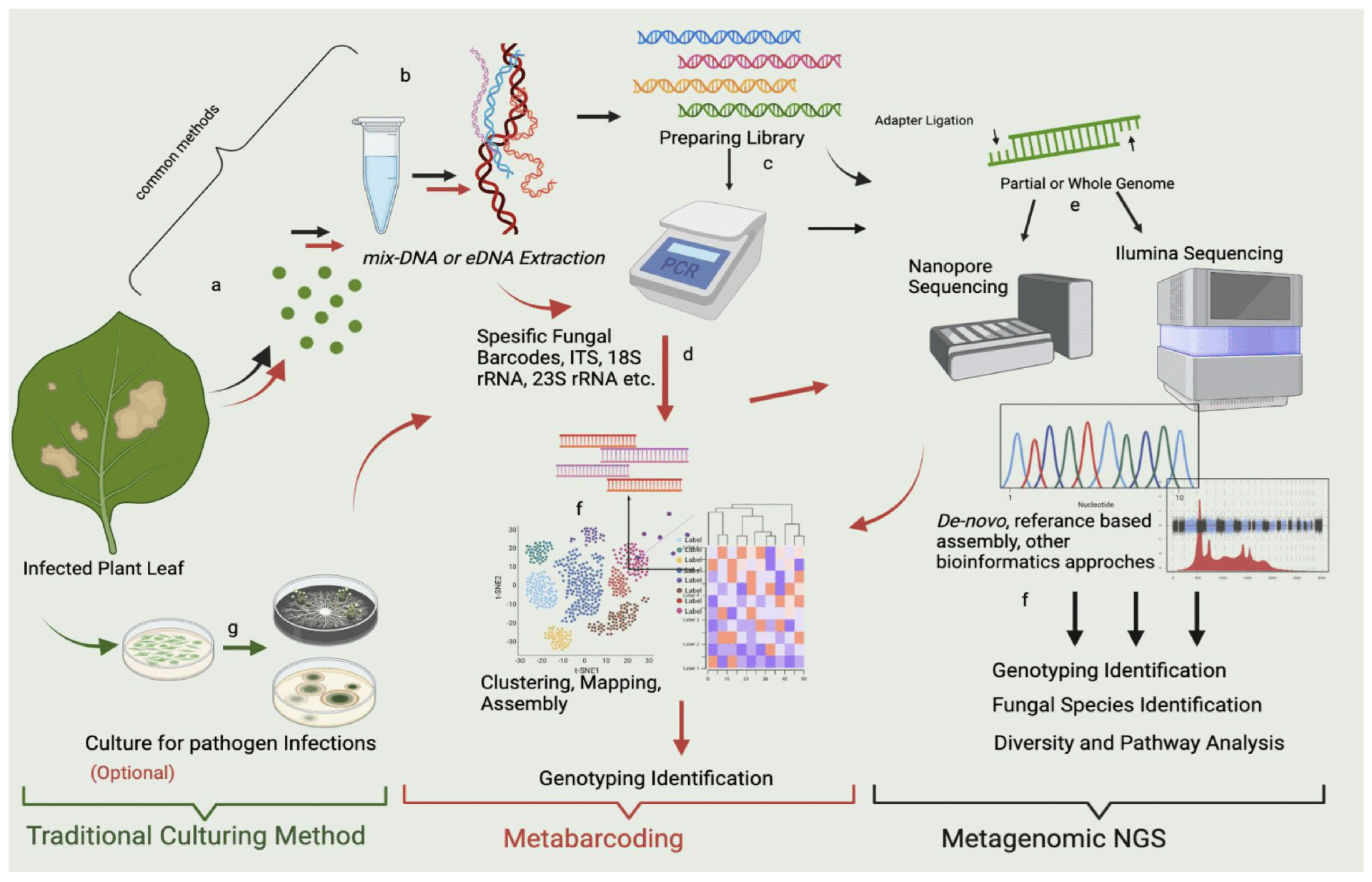
What mNGS Is and Isn’t ?
mNGS is hypothesis-free detection : instead of testing for a narrow set of targets, it sequences total (or enriched) nucleic acids from the specimen and classifies reads taxonomically. It is not a replacement for all targeted assays or culture; rather, it complements them when the etiology is unclear, mixed, or rare, or when prior antimicrobials suppress culture yield.
- Culture-negative infections: endocarditis, osteomyelitis, prosthetic joint infections, or prior-antibiotic exposure.
- Atypical or mixed infections: polymicrobial abscesses, complicated pneumonia, intra-abdominal sepsis.
- Immunocompromised hosts: broad search across opportunistic bacteria, viruses (DNA/RNA), and fungi.
- Rare or emerging pathogens: zoonoses, travel-related infections, or outbreaks with unknown etiology.
Specimen Types & Pre-Analytics (High Impact Area)
- Common matrices: whole blood, plasma/serum, CSF, BAL/sputum, tissue/abscess aspirate, stool, swabs, sterile fluids.
- Stabilization: immediate cooling or nucleic-acid stabilization; avoid freeze–thaw.
- Contamination control: clean collection, low-biomass safeguards, lot-tracked reagents, physical separation of pre-/post-PCR areas.
- Metadata capture: collection time, prior antimicrobials, suspected focus, travel/exposure risks critical for interpretation.
- Extraction & QC – Input normalization; internal extraction control; DNA/RNA workflows as indicated.
- Optional host depletion – Physical/enzymatic depletion to increase microbial signal (matrix-dependent).
- Library preparation – Fragmentation, adapter/index ligation, clean-ups; negative/positive controls included.
- Sequencing – Short-read platform with run-level controls to monitor carryover/contamination.
- Read QC : Adapter trimming, quality filtering, deduplication.
- Host subtraction : Alignment to the host genome to reduce background.
- Taxonomic classification : K-mer/classifier or alignment-based calls against curated databases (bacteria, viruses, fungi, parasites).
- Post-processing : Decontamination filters, complexity checks, and background modeling using NTCs and environmental controls.
- Quantification : Raw reads and normalized abundance (e.g., reads per million, genome coverage/ breadth).
- Confidence scoring : Thresholding by read depth, coverage patterns, uniqueness of mapped regions, and replicate support.
- Versioning matters : lock database versions, classifier parameters, and pipeline code; record them in the report.
- Matrix & clinical concordance: Does the organism plausibly explain the syndrome and specimen type?
- Signal strength: Reads, normalized abundance, and coverage uniformity vs. spurious low-complexity hits.
- Controls: Absent/present in NTCs? Any batch-specific reagent contaminants?
- Co-detections: Distinguish colonizers/commensals from pathogens (especially in respiratory and stool).
- Actionability & next steps: Need confirmatory testing (culture, targeted PCR, antigen/serology) or source control?
- Run-level NTCs and extraction blanks processed in parallel.
- Cross-run background database of recurrent contaminants.
- Thresholds that adapt to matrix (e.g., CSF vs. BAL) and library complexity.
- Lot traceability for kits/reagents; environmental swabs for incident review.
- Analytical sensitivity (LoD): Varies by organism, matrix, extraction efficiency, host depletion, library complexity, and sequencing depth. Validate per matrix.
- Specificity: Curated reference databases, stringent classifier settings, and robust background models are essential.
- Turnaround time: Panels can be same day; mNGS is typically ~24–48 h from extraction to report depending on batching and depth.
- Quantitation caveats: Read counts reflect nucleic-acid burden, not necessarily viable organisms or susceptibility
- Targeted multiplex panels: Rapid, sensitive for known agents; limited to predefined targets.
- mNGS: Broad, hypothesis-free discovery; ideal for unknown, mixed, or rare etiologies and post-panel escalation.
- Pragmatic pathway: Start with a high-yield panel; escalate to mNGS for unresolved, severe, or atypical cases.
Unbiased detection across all major pathogen classes in a single assay, especially valuable when routine tests are negative or the presentation is atypical.
No. Culture remains essential for antimicrobial susceptibility testing and epidemiology. mNGS complements culture and targeted assays.
The ranked organism list, normalized abundance, and coverage inform likelihood of true infection; QC and contamination screening set confidence; interpretation notes connect findings to the clinical picture.
With streamlined workflows, 24–48 hours is common from extraction to report; urgent pathways may shorten analysis at the expense of depth.
Strict pre-analytics, comprehensive controls, curated databases, and matrix-aware thresholds reduce both; results should be integrated with clinical data and when needed confirmed.
Metagenomic Pathogen Detection (mNGS)
mNGS turns complex, ambiguous infection workups into data-driven differentials by delivering a transparent, QC-anchored report: ranked taxa, read and coverage metrics, contamination screens, and interpretation notes. Used alongside targeted panels, culture, and imaging, it closes diagnostic gaps in culture-negative, atypical, mixed, and immunocompromised infections precisely where conventional workflows struggle.